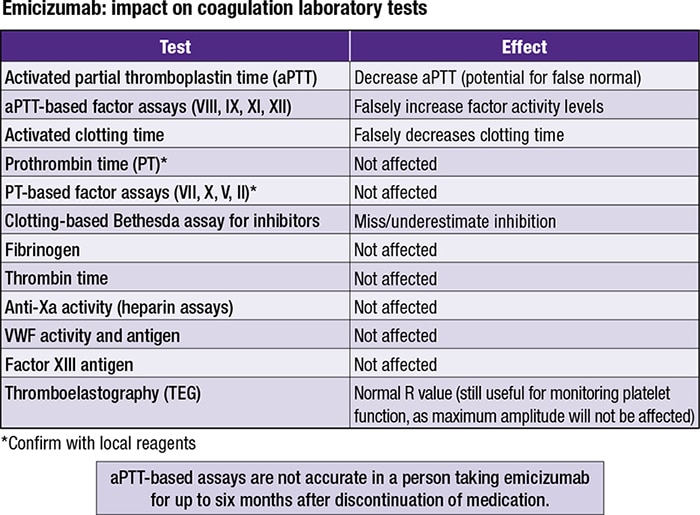
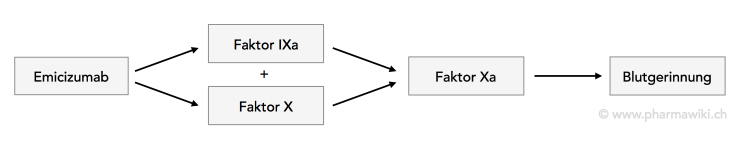
Valoctocogene Roxaparvovec and Emicizumab for Hemophilia A without Inhibitors: Effectiveness and Value

Arun KEEPANASSERIL | Research and Data Operations Lead, McMaster Hemophilia Resrarch Group | MDS,MSc,CPHIMS -CA | McMaster University, Hamilton | McMaster | Health Information Research Unit (HIRU) | Research profile
Simoctocog Alfa (Nuwiq) in Previously Untreated Patients with Severe Haemophilia A: Final Results of the NuProtect Study

AKATSUKI Study: A Prospective, Multicenter, Phase IV Study to Evaluate the Safety of Emicizumab Under and Immediately After Immune Tolerance Induction (ITI) Therapy in Persons with Congenital Hemophilia A (PwHA) with Factor (

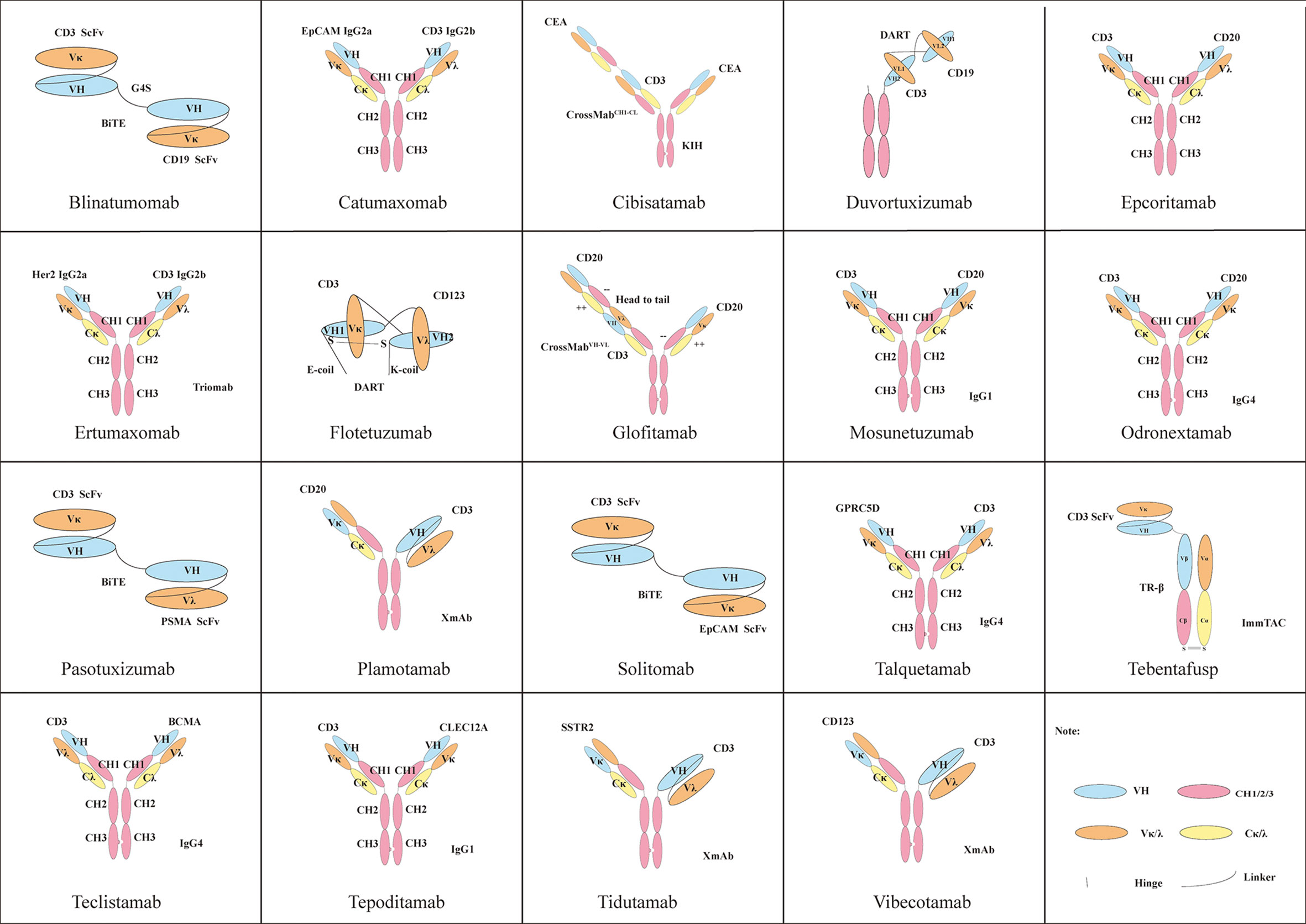
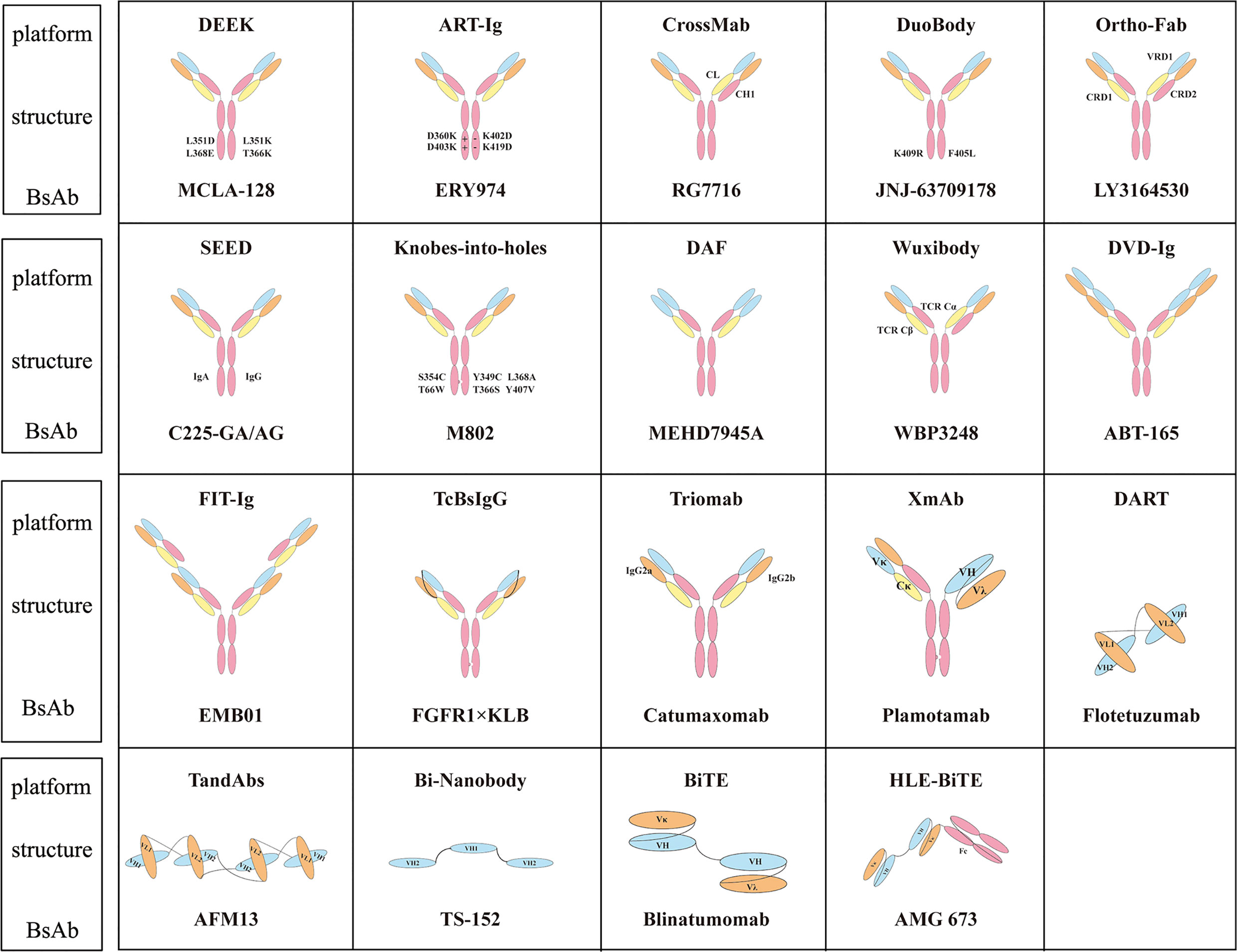
An anti–glypican 3/CD3 bispecific T cell–redirecting antibody for treatment of solid tumors | Science Translational Medicine